Catalytically active ceria-supported cobalt–manganese oxide nanocatalysts for oxidation of carbon monoxide†
Abstract
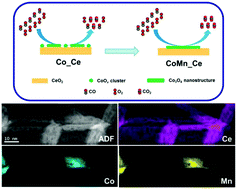
A low-concentration cobalt (∼6 at%) and manganese (∼3 at%) bimetallic oxide catalyst supported on ceria nanorods (CoMnOx/CeO2), as well as its related single metal oxide counterparts (CoOx/CeO2 and MnOx/CeO2) was synthesized via a deposition–precipitation approach. The fresh samples after air-calcination at 400 °C were tested under the reaction conditions of CO oxidation, and showed the following order of reactivity: CoMnOx/CeO2 > CoOx/CeO2 > MnOx/CeO2. X-ray diffraction (XRD) and transmission electron microscopy (TEM) data identified that the structure of the CeO2 support was maintained during deposition of metal (Co, Mn) ions while the corresponding vis-Raman spectra verified that more oxygen vacancies were created after deposition–precipitation than those in pure ceria nanorods. Aberration-corrected, high-angle, annular dark-field scanning transmission electron microscopy (HAADF-STEM) images with the help of electron energy loss spectroscopy (EELS) analyses determined two types of cobalt species, i.e. ultra-fine clusters (<2 nm) and smaller nanocrystals (up to 5 nm) in CoOx/CeO2 while only bigger nanostructures (∼10 nm) of cobalt–manganese oxides in CoMnOx/CeO2. X-ray absorption fine structure (XAFS) measurements demonstrated the presence of a cubic Co3O4 phase in all the cobalt-based catalysts. The fitting results of the extended X-ray absorption fine structure (EXAFS) indicated that the introduction of the secondary metal (Mn) oxide significantly enhanced the two-dimensional growth of cobalt oxide nanostructures on the surface of CeO2. Therefore, the enhanced activity of CO oxidation reaction over the bimetallic cobalt–manganese oxide nanocatalyst can be attributed to the higher crystallinity of the Co3O4 phase in this work.


 Please wait while we load your content...
Please wait while we load your content...