The role of tachysterol in vitamin D photosynthesis – a non-adiabatic molecular dynamics study†
Abstract
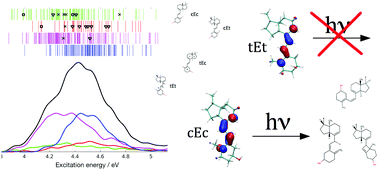
To investigate the role of tachysterol in the photophysical/photochemical regulation of vitamin D photosynthesis, we studied its electronic absorption properties and excited state dynamics using time-dependent density functional theory (TDDFT), second-order approximate coupled cluster theory (CC2), and non-adiabatic surface hopping molecular dynamics in the gas phase. In excellent agreement with experiments, the simulated electronic spectrum shows a broad absorption band with a remarkably higher extinction coefficient than the other vitamin D photoisomers provitamin D, lumisterol, and previtamin D. The broad band arises from the spectral overlap of four different ground state rotamers. After photoexcitation, the first excited singlet state (S1) decays with a lifetime of 882 fs. The S1 dynamics is characterized by a strong twisting of the central double bond. In 96% of all trajectories this is followed by unreactive relaxation to the ground state near a conical intersection. The double-bond twisting in the chemically unreactive trajectories induces a strong interconversion between the different rotamers. In 2.3% of the trajectories we observed [1,5]-sigmatropic hydrogen shift forming the partly deconjugated toxisterol D1. 1.4% previtamin D formation is observed via hula-twist double bond isomerization. In both reaction channels, we find a strong dependence between photoreactivity and dihedral angle conformation: hydrogen shift only occurs in cEc and cEt rotamers and double bond isomerization occurs mainly in cEc rotamers. Hence, our study confirms the previously formed hypothesis that cEc rotamers are more prone to previtamin D formation than other isomers. In addition, we also observe the formation of a cyclobutene-toxisterol in the hot ground state in 3 trajectories (0.7%). Due to its large extinction coefficient and mostly unreactive behavior, tachysterol acts mainly as a Sun shield suppressing previtamin D formation. Tachysterol shows stronger toxisterol formation than previtamin D and can thus be seen as the major degradation route of vitamin D. Absorption of low energy ultraviolet light by the cEc rotamer can lead to previtamin D formation. In addition, the cyclobutene-toxisterol, which possibly reacts thermally to previtamin D, is also preferably formed at long wavelengths. These two mechanisms are consistent with the wavelength dependent photochemistry found in experiments. Our study reinforces a recent hypothesis that tachysterol constitutes a source of previtamin D when only low energy ultraviolet light is available, as it is the case in winter or in the morning and evening hours of the day.
- This article is part of the themed collection: 2017 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...