Exploring the relevance of gas-phase structures to biology: cold ion spectroscopy of the decapeptide neurokinin A†
Abstract
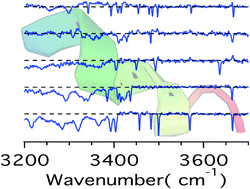
Linking the intrinsic tertiary structures of biomolecules to their native geometries is a central prerequisite for making gas-phase studies directly relevant to biology. The isolation of molecules in the gas phase eliminates hydrophilic interactions with solvents, to some extent mimicking a hydrophobic environment. Intrinsic structures therefore may resemble native ones for peptides that in vivo reside in a hydrophobic environment (e.g., binding pockets of receptors). In this study, we investigate doubly protonated neurokinin A (NKA) using IR-UV double resonance cold ion spectroscopy and find only five conformers of this decapeptide in the gas phase. In contrast, NMR data show that in aqueous solutions, NKA exhibits high conformational heterogeneity, which reduces to a few well-defined structures in hydrophobic micelles. Do the gas-phase structures of NKA resemble these native structures? The IR spectra reported here allow the validation of future structural calculations that may answer this question.


 Please wait while we load your content...
Please wait while we load your content...