Eu3+-Doped Y3−xNdxAl3O12 garnet: synthesis and structural investigation†
Abstract
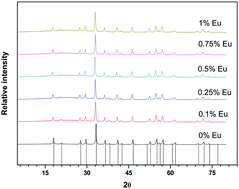
Nd3+-Doped yttrium aluminium garnet and Eu3+–Nd3+-co-doped yttrium aluminium garnet were synthesized using an environmentally friendly sol–gel method at low temperatures. The results of X-ray diffraction (XRD) analysis of the powders sintered at 1000 °C showed the purity and formation of monophasic compounds. The phase composition and purity confirmation of the samples were also characterized via FTIR spectroscopy. Desirable microstructural features and particle size of phosphorous materials of the polycrystalline samples were studied via scanning electron microscopy (SEM). The local environments of Eu3+ and Nd3+ activator ions of the garnets structure compounds were investigated using nuclear magnetic resonance (NMR) spectroscopy. The local environments of the small number of substituted phosphorous ions (Eu3+ and Nd3+) in YAG were shown to critically influence optical properties. Structural features of garnets were found to correlate with their luminescence properties. The luminescence properties were characterized using the results from a photoluminescence (PL) study. Effective concentration of luminescent ion – Nd in YAG was, observed and the optimum ratio between two phosphorous (Eu3+ and Nd3+) was analysed. The transition among europium and neodymium phosphorus was determined.


 Please wait while we load your content...
Please wait while we load your content...