OH production from the photolysis of isoprene-derived peroxy radicals: cross-sections, quantum yields and atmospheric implications†
Abstract
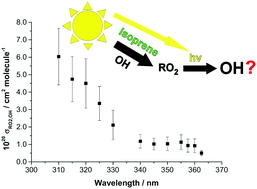
In environments with high concentrations of biogenic volatile organic compounds and low concentrations of nitrogen oxides (NOx = NO + NO2), significant discrepancies have been found between measured and modeled concentrations of hydroxyl radical (OH). The photolysis of peroxy radicals from isoprene (HO-Iso-O2) in the near ultraviolet represents a potential source of OH in these environments, yet has not been considered in atmospheric models. This paper presents measurements of the absorption cross-sections for OH formation (σRO2,OH) from the photolysis of HO-Iso-O2 at wavelengths from 310–362.5 nm, via direct observation by laser-induced fluorescence of the additional OH produced following laser photolysis of HO-Iso-O2. Values of σRO2,OH for HO-Iso-O2 ranged from (6.0 ± 1.6) × 10−20 cm2 molecule−1 at 310 nm to (0.50 ± 0.15) × 10−20 cm2 molecule−1 at 362.5 nm. OH photodissociation yields from HO-Iso-O2 photolysis, ϕOH,RO2, were determined via comparison of the measured values of σRO2,OH to the total absorption cross-sections for HO-Iso-O2 (σRO2), which were obtained using a newly-constructed spectrometer. ϕOH,RO2 was determined to be 0.13 ± 0.04 at wavelengths from 310–362.5 nm. To determine the impact of HO-Iso-O2 photolysis on atmospheric OH concentrations, a modeling case-study for a high-isoprene, low-NOx environment (namely, the 2008 Oxidant and Particle Photochemical Processes above a South-East Asian Tropical Rainforest (OP-3) field campaign, conducted in Borneo) was undertaken using the detailed Master Chemical Mechanism. The model calculated that the inclusion of HO-Iso-O2 photolysis in the model had increased the OH concentration by only 1% on average from 10:00–16:00 local time. Thus, HO-Iso-O2 photolysis alone is insufficient to resolve the discrepancy seen between measured OH concentrations and those predicted by atmospheric chemistry models in such environments.
- This article is part of the themed collection: Celebrating the 2017 RSC Prize and Award Winners


 Please wait while we load your content...
Please wait while we load your content...