Understanding dispersive charge-transport in crystalline organic-semiconductors†
Abstract
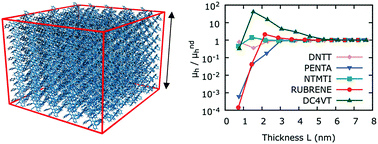
The effect of short-range order and dispersivity on charge-transport for organic crystalline semiconductors are important and unresolved questions. This exploration is the first to discern the role of short-range order on charge-transport for crystalline organic semiconductors. A multimode computational approach (including Molecular Dynamics and kinetic Monte Carlo simulations) is employed to understand the hole mobility dispersivity of crystalline organic semiconductors. Crystalline organic solids feature a mesoscale region where dispersive charge-transport dominates; our calculations show a clear transition of charge-mobility from non-dispersive to dispersive. An empirical relationship between the dispersive and non-dispersive transport transition region and ideal simulation box thickness is put forth. The dispersive to non-dispersive transition region occurs when energetic disorder approaches 72 meV. Non-dispersive transport is observed for simulation box sizes greater than 3.7 nm, which corresponds to approximately 12 π-stacked layers in typical π-stacked organic solids. A qualitative relationship is deduced between the variability of measured dispersive hole and variability of computed dispersive hole mobilities and system size. This relationship will guide future charge-transport investigations of condensed-phase organic systems.

 Please wait while we load your content...
Please wait while we load your content...