Incorporation of molybdenum(vi) in akaganéite (β-FeOOH) and the microbial reduction of Mo–akaganéite by Shewanella loihica PV-4†
Abstract
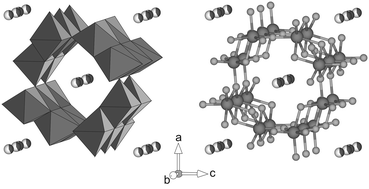
Among all highly-crystalline iron oxides present in the environment, akaganéite (β-FeO(OH, Cl)) possesses one of the most unconventional structural setups and is a known scavenger for large quantitates of molybdenum (Mo6+). The factors controlling the exact mechanism for Mo6+ incorporation into the akaganéite crystal structure are poorly understood and the ability of dissimilatory Fe(III)-reducing microorganisms to reduce pure akaganéite or Mo-carrying iron oxides is not well characterized. In the current study, we investigated the short-range order around Mo6+ in akaganéite and the fate of Mo6+ under microbially-mediated Fe(III)-reducing conditions. We found that akaganéite can incorporate up to 14.11 ± 0.22 wt% Mo, while the Fe content decreases from 59.70 ± 0.31 to 40.40 ± 0.24 wt%, which indicates a loss of 2–3 Fe atoms for each Mo incorporated. Simultaneously, the crystal structure unit cell parameters a, b and c decrease, while β increases with increasing Mo content. Surprisingly, dissolution of akaganéite by Shewanella loihica PV-4 showed higher dissolution rates of Mo-bearing akaganéite compared to Mo-free akaganéite. Moreover, these results suggest the reduction of Mo6+ is most likely microbially-induced (Fe3+ → Fe2+, Mo6+ + 2Fe2+ → Mo4+ + 2Fe3+). Furthermore, X-ray absorption spectra collected at the Mo L3-edge show a peak-splitting of the white line with a splitting gap of 2.7 eV and an increased amplitude for the first peak. This observation indicates Mo6+ is octahedrally coordinated by oxygen, assuming a strongly distorted MoO6-octhaedron. Fitting of the short-range order around Mo6+ in akaganéite supports the presence of a strongly distorted MoO6-octahedron in a coordination environment similar to the Fe position in akaganéite and the formation of Fe-vacancies close to the newly incorporated Mo6+.


 Please wait while we load your content...
Please wait while we load your content...