Co-crystallization of 1,3,5-trifluoro-2,4,6-triiodobenzene (1,3,5-TFTIB) with a variety of Lewis bases through halogen-bonding interactions
Abstract
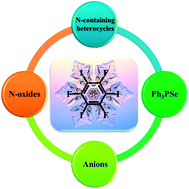
The halogen bond is emerging as an important driving force for supramolecular self-assembly, and has attracted great interest in the past decade. Among the ample halogen-bonding donors, we note the ability of 1,3,5-trifluoro-2,4,6-triiodobenzene (1,3,5-TFTIB) to co-crystallize with various halogen-bonding acceptors, ranging from neutral Lewis bases (nitrogen-containing heterocycles, N-oxides, and triphenylphosphine selenide (Ph3PSe)) to anions (halide ions and thiocyanate ion), leading to a wide diversity of supramolecular architectures. Some of them are promising optoelectronic functional materials. In this review, we concentrate on the structures of multicomponent supramolecular complexes, highlight typical examples, and point out the main possible directions that remain to be developed in this field. From the perspectives of supramolecular chemistry and crystal engineering, the complexes reviewed here should provide useful information for further design and investigation on this fascinating class of halogen-bonding supramolecular architectures.
- This article is part of the themed collection: 2017 Highlight article collection


 Please wait while we load your content...
Please wait while we load your content...