Fluorination promotes chalcogen bonding in crystalline solids†
Abstract
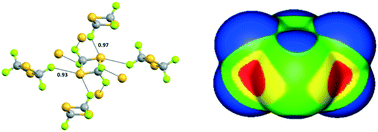
Single crystal X-ray analysis shows that intermolecular C–S⋯F chalcogen bonds are by far the shortest contacts in the crystal structure of 2,2,4,4-tetrafluoro-1,3-dithietane. Analysis of the molecular surface electrostatic potentials indicates that these contacts result from the attraction between the negative potentials of fluorines and the positive regions resulting from the overlapping of sulfur and carbon positive σ-holes. In contrast, the lattice structure of 2,2,4,4-tetrachloro-1,3-dithietane is controlled by intermolecular C–Cl⋯Cl halogen bonds. The lattices of the two systems are thus controlled by two different σ-hole interactions.
- This article is part of the themed collection: 1st International Conference on Noncovalent Interactions


 Please wait while we load your content...
Please wait while we load your content...