Endo-/exo- and halogen-bonded complexes of conformationally rigid C-ethyl-2-bromoresorcinarene and aromatic N-oxides†
Abstract
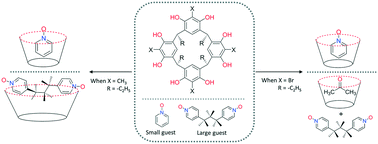
The host–guest complexes of conformationally rigid C-ethyl-2-bromoresorcinarene with aromatic N-oxides were studied using single crystal X-ray crystallography. Unlike that of the conformationally more flexible C-ethyl-2-methylresorcinarene, the C-ethyl-2-bromoresorcinarene cavity forms endo-complexes only with the small pyridine-N-oxides, such as pyridine N-oxide, 2-methyl-, 3-methyl- and 4-methylpyrdine N-oxide, and quinoline N-oxide. The larger 2,4,6-trimethylpyridine, 4-phenylpyridine and isoquinoline N-oxide, and 4,4-bipyridine N,N′-dioxide and 1,3-bis(4-pyridyl)propane N,N′-dioxide do not fit into the host cavity. Instead endo-acetone complexes are formed. Remarkably, differing from the anti–gauche endo-complex with C-ethyl-2-methylresorcinarene, the flexible 1,3-bis(4-pyridyl)propane N,N′-dioxide guest forms an anti–anti exo-complex with C-ethyl-2-bromoresorcinarene. The endo- and exo-complexes of C-ethyl-2-bromoresorcinarene and studied N-oxides manifest C–O⋯Br, C–H⋯π and C–Br⋯π interactions.
- This article is part of the themed collections: Host‒Guest chemistry: in honour of Luigi Nassimbeni’s 9th decade and 1st International Conference on Noncovalent Interactions


 Please wait while we load your content...
Please wait while we load your content...