Experimental and simulation-based understanding of morphology controlled barium titanate nanoparticles under co-adsorption of surfactants
Abstract
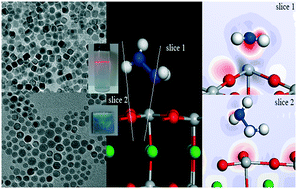
Well dispersed single-crystalline BaTiO3 nanoparticles with controlled morphologies were synthesized using a thermohydrolysis route. The nanoparticles were tuned from spherical to cubic upon changing the NaOH concentration under a critical molar ratio of oleic acid to hydrazine. Density functional theory (DFT) and molecular dynamics (MD) calculations indicated that hydrazine molecules adsorbed preferably on the Ti position of the Ti–O terminated surface; meanwhile, oleic acid molecules tended to adsorb on the Ba position of the Ba–O terminated surface. The added hydrazine changed the formation mechanism of BaTiO3 nanoparticles from an in situ growth to a dissolution–precipitation growth. Excellent dispersibility in aqueous solution was achieved for the BaTiO3 nanoparticles under the assistance of hydrazine. Meanwhile, a high-quality self-assembled film with a stable dielectric constant of 30 in the frequency range from 0 Hz to 1 MHz was prepared using the well dispersed BaTiO3 nanoparticles, providing a novel low-temperature route for the fabrication of perovskite films.
- This article is part of the themed collection: A celebration of 25 volumes of CrystEngComm


 Please wait while we load your content...
Please wait while we load your content...