Chiral phosphoric acid-catalyzed desymmetrizative glycosylation of 2-deoxystreptamine and its application to aminoglycoside synthesis†
Abstract
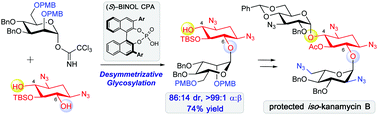
This work describes chiral phosphoric acid (CPA)-catalyzed desymmetrizative glycosylation of meso-diol derived from 2-deoxystreptamine. The chirality of CPA dictates the outcome of the glycosylation reactions, and the use of enantiomeric CPAs results in either C4-glycosylated (67 : 33 d.r.) or C6-glycosylated (86 : 14 d.r.) 2-deoxystreptamines. These glycosylated products can be converted to aminoglycosides, and the application of this strategy to the synthesis of protected iso-neamine and iso-kanamycin B with inverted connection at the C4 and C6 positions is described.
- This article is part of the themed collection: Site-selective molecular transformations


 Please wait while we load your content...
Please wait while we load your content...