Carbodiimide insertion into sulfonimides: one-step route to azepine derivatives via a two-atom saccharin ring expansion†
Abstract
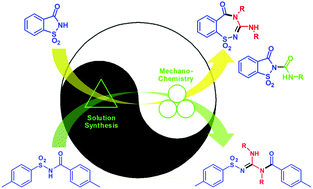
A previously unknown insertion of carbodiimides into sulfonimides enables the first one-step, two-atom expansion of the saccharin 5-membered ring into a 7-membered benzo[1,2,4]thiadiazepine, and a two-atom chain extension of a non-cyclic sulfonimide. This reaction is enhanced by copper salts, which allow it to be conducted mechanochemically, in a solvent-free manner.
- This article is part of the themed collection: Celebrating the ChemComm Emerging Investigator Lectureship Winners


 Please wait while we load your content...
Please wait while we load your content...