Bioreducible cross-linked core polymer micelles enhance in vitro activity of methotrexate in breast cancer cells†
Abstract
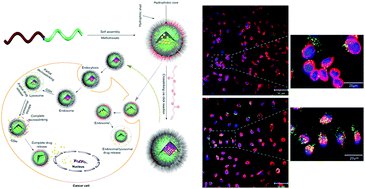
Polymer micelles have emerged as promising carriers for controlled release applications, however, several limitations of micelle-based drug delivery have also been reported. To address these issues, we have synthesized a functional biodegradable and cytocompatible block copolymer based on methoxypoly(ethyleneglycol)-b-poly(ε-caprolactone-co-α-azido-ε-caprolactone) (mPEG-b-poly(εCL-co-αN3εCL)) as a precursor of reduction sensitive core-crosslinked micelles. The synthesized polymer was formulated as micelles using a dialysis method and loaded with the anti-inflammatory and anti-cancer drug methotrexate (MTX). The micellar cores were subsequently crosslinked at their pendent azides by a redox-responsive bis(alkyne). The size distributions and morphology of the polymer micelles were assessed using dynamic light scattering (DLS) and transmission electron microscopy, and drug release assays were performed under simplified (serum free) physiological and reductive conditions. Cellular uptake studies in human breast cancer cells were performed using Oregon-green loaded core-crosslinked micelles. The MTX-loaded core-crosslinked micelles were assessed for their effects on metabolic activity in human breast cancer (MCF-7) cells by evaluating the reduction of the dye MTT 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide. The apoptosis inducing potential of MTX-loaded core-crosslinked micelles was analysed using Hoechst/propidium iodide (PI) and annexin-V/PI assays. The data from these experiments indicated that drug release from these cross-linked micelles can be controlled and that the redox-responsive micelles are more effective carriers for MTX than non-crosslinked analogues and the free drug in the cell-lines tested.

 Please wait while we load your content...
Please wait while we load your content...