Improving gelation efficiency and cytocompatibility of visible light polymerized thiol-norbornene hydrogels via addition of soluble tyrosine†
Abstract
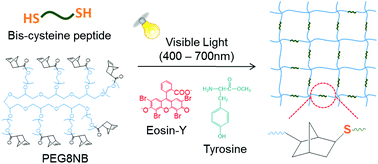
Hydrogels immobilized with biomimetic peptides have been used widely for tissue engineering and drug delivery applications. Photopolymerization has been among the most commonly used techniques to fabricate peptide-immobilized hydrogels as it offers rapid and robust peptide immobilization within a crosslinked hydrogel network. Both chain-growth and step-growth photopolymerizations can be used to immobilize peptides within covalently crosslinked hydrogels. A previously developed visible light mediated step-growth thiol-norbornene gelation scheme has demonstrated efficient crosslinking of hydrogels composed of an inert poly(ethylene glycol)-norbornene (PEGNB) macromer and a small molecular weight bis-thiol linker, such as dithiothreitol (DTT). Compared with conventional visible light mediated chain-polymerizations where multiple initiator components are required, step-growth photopolymerized thiol-norbornene hydrogels are more cytocompatible for the in situ encapsulation of radical sensitive cells (e.g., pancreatic β-cells). This contribution explored visible light based crosslinking of various bis-cysteine containing peptides with macromer 8-arm PEGNB to form biomimetic hydrogels suitable for in situ cell encapsulation. It was found that the addition of soluble tyrosine during polymerization not only significantly accelerated gelation, but also improved the crosslinking efficiency of PEG-peptide hydrogels as evidenced by a decreased gel point and enhanced gel modulus. In addition, soluble tyrosine drastically enhanced the cytocompatibility of the resulting PEG-peptide hydrogels, as demonstrated by in situ encapsulation and culture of pancreatic MIN6 β-cells. This visible light based thiol-norbornene crosslinking mechanism provides an attractive gelation method for preparing cytocompatible PEG-peptide hydrogels for tissue engineering applications.

 Please wait while we load your content...
Please wait while we load your content...