Selective and low potential electrocatalytic oxidation and sensing of l-cysteine using metal impurity containing carbon black modified electrode†
Abstract
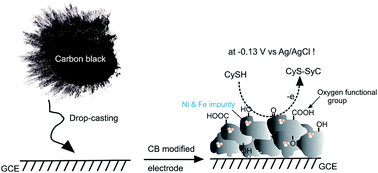
The detection of thiol-containing amino acids, particularly L-cysteine (L-CySH), without any interference from other biochemicals is a challenging research interest in electroanalytical chemistry. Amongst various electrodes, the nanogold modified electrode has been reported to be effective for low potential electrochemical oxidation (∼0 V vs. Ag/AgCl) and sensing of L-CySH. Herein, we report a conductive carbon black (CB, CL-08) modified glassy carbon electrode (GCE/CL-08) with a high surface area (1000 m2 g−1), prepared by a simple drop-casting technique for efficient electrocatalytic oxidation at the lowest oxidation potential, −0.13 V vs. Ag/AgCl, and sensing of L-CySH in a pH 6.65 phosphate buffer solution. The trace metal impurities such as Ni (0.18 wt%) and Fe (0.42 wt%) in CL-08 are found to be key for such unique and unusual electrocatalytic property observed in this study. Compared with the multiwalled carbon nanotube and graphite powder modified electrodes, the GCE/CL-08 showed about 400 mV reduction in the oxidation potential and twenty times enhancement in the current signal for the CySH. A chronoamperometry detection of L-CySH on the GCE/CL-08 at an applied potential = −0.13 V vs. Ag/AgCl yielded a current linearity from 50 to 700 μM with a LOD = 45.87 nM. There are no interferences by common biochemicals such as ascorbic acid, dopamine, uric acid, xanthine, hypoxanthine and homo-cysteine on the L-CySH oxidation potential. Selective chronoamperometric detection of L-CySH in the blood serum demonstrated ∼100% recovery value as a validation for the present protocol.


 Please wait while we load your content...
Please wait while we load your content...