A squaraine-based sensor for colorimetric detection of CO2 gas in an aqueous medium through an unexpected recognition mechanism: experiment and DFT calculation†
Abstract
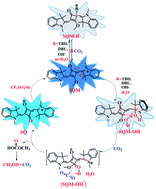
A cationic squaraine-based chemosensor SQM was synthesized and its sensing behaviors towards nucleophiles followed by CO2 gas were described in detail by interpreting the results from UV-Vis, ESI-MS, and 1H NMR spectral analysis. The results indicated that there were two different paths for the bleaching of SQM, depending on whether water existed or not when the nucleophilic addition reaction between SQM and a superbase, such as TBD or DBU, occurred. More interestingly, the bleached SQM in MeCN–H2O (v/v = 90 : 10) could be colorimetrically restored with bubbling CO2 gas, which enabled it to act as a highly sensitive “naked-eye” CO2 gas detector in an aqueous medium. However, when treated in the same way, the SQM bleached in MeCN remained unchanged, unexpectedly. Combining theoretical analysis and experimental tests, a plausible sensing mechanism was proposed to illustrate the bleaching of SQM and the response of the bleached SQM to CO2 gas in an aqueous medium.


 Please wait while we load your content...
Please wait while we load your content...