Tridentate tripodal sulfur ligand as a stable molecular surface anchor for the fabrication of oligonucleotide-gold based label-free biosensors
Abstract
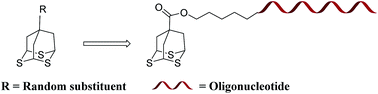
The syntheses of a novel tripodal surface anchor, 2,4,9-trithiaadamantane, are reported. The rigid, highly symmetric, tridentate nature of the tripod thioether, with less susceptibility to oxidation, and temperature fluctuations, makes it a highly suitable surface anchor for the attachment of biomolecules onto gold surfaces for bioanalytical purposes. We used tridentate sulfur ligands, modified with a phosphoramidite end group, to develop stable oligonucleotide-functionalized gold nanoparticles, exhibiting similar hybridization properties when compared to previously reported alkane thiol anchors. The stability experiments showed enhanced resistance of the tripod oligonucleotide probes towards exchange reactions with the thiols in solution. UV-vis and surface plasmon resonance (SPR) spectroscopies were further used to demonstrate the potential of the tripod as a molecular surface anchor for the detection of free target oligonucleotides, displaying a promising approach towards the development of label-free oligonucleotide based biosensors.


 Please wait while we load your content...
Please wait while we load your content...