An Aptamer Bio-barCode (ABC) assay using SPR, RNase H, and probes with RNA and gold-nanorods for anti-cancer drug screening
Abstract
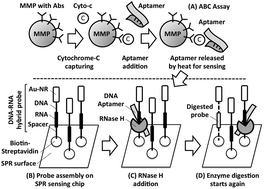
With modifications to an ultra-sensitive bio-barcode (BBC) assay, we have developed a next generation aptamer-based bio-barcode (ABC) assay to detect cytochrome-c (Cyto-c), a cell death marker released from cancer cells, for anti-cancer drug screening. An aptamer is a short single-stranded DNA selected from a synthetic DNA library that is capable of binding to its target with high affinity and specificity based on its unique DNA sequence and 3D structure after folding. Similar to the BBC assay, Cyto-c is captured by a micro-magnetic particle (MMP) coated with capturing antibodies (Ab) and an aptamer specifically against Cyto-c to form sandwich structures ([MMP-Ab]–[Cyto-c]–[Aptamer]). After washing and melting, our aptamers, acting as a DNA bio-barcode, are released from the sandwiches and hybridized with the probes specially designed for RNase H for surface plasmon resonance (SPR) sensing. In an aptamer–probe duplex, RNase H digests the RNA in the probe and releases the intact aptamer for another round of hybridization and digestion. With signal enhancement effects from gold-nanorods (Au-NRs) on probes for SPR sensing, the detection limit was found to be 1 nM for the aptamer and 80 pM for Cyto-c. Without the time-consuming DNA amplification steps by PCR, the detection process of this new ABC assay can be completed within three hours. As a proof-of-concept, phenylarsine oxide was found to be a potent agent to kill liver cancer cells with multi-drug resistance at the nano-molar level. This approach thus provides a fast, sensitive and robust tool for anti-cancer drug screening.


 Please wait while we load your content...
Please wait while we load your content...