Quantifying spontaneous metastasis in a syngeneic mouse melanoma model using real time PCR†
Abstract
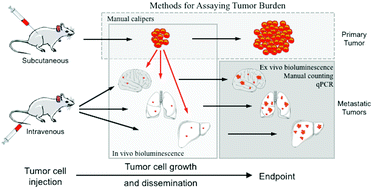
Modeling metastasis in vivo with animals is a priority for both revealing mechanisms of tumor dissemination and developing therapeutic methods. While conventional intravenous injection of tumor cells provides an efficient and consistent system for studying tumor cell extravasation and colonization, studying spontaneous metastasis derived from orthotopic tumor sites has the advantage of modeling more aspects of the metastatic cascade, but is challenging as it is difficult to detect small numbers of metastatic cells. In this work, we developed an approach for quantifying spontaneous metastasis in the syngeneic mouse B16 system using real time PCR. We first transduced B16 cells with lentivirus expressing firefly luciferase Luc2 gene for bioluminescence imaging. Next, we developed a real time quantitative PCR (qPCR) method for the detection of luciferase-expressing, metastatic tumor cells in mouse lungs and other organs. To illustrate the approach, we quantified lung metastasis in both spontaneous and experimental scenarios using B16F0 and B16F10 cells in C57BL/6Ncrl and NOD-Scid Gamma (NSG) mice. We tracked B16 melanoma metastasis with both bioluminescence imaging and qPCR, which were found to be self-consistent. Using this assay, we can quantitatively detect one Luc2 positive tumor cell out of 104 tissue cells, which corresponds to a metastatic burden of 1.8 × 104 metastatic cells per whole mouse lung. More importantly, the qPCR method was at least a factor of 10 more sensitive in detecting metastatic cell dissemination and should be combined with bioluminescence imaging as a high-resolution, end-point method for final metastatic cell quantitation. Given the rapid growth of primary tumors in many mouse models, assays with improved sensitivity can provide better insight into biological mechanisms that underpin tumor metastasis.


 Please wait while we load your content...
Please wait while we load your content...