Monitoring ATPase induced pH changes in single proteoliposomes with the lipid-coupled fluorophore Oregon Green 488†
Abstract
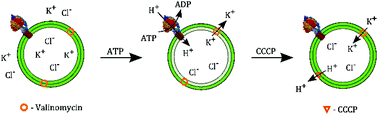
Monitoring the proton pumping activity of proteins such as ATPases in reconstituted single proteoliposomes is key to quantify the function of proteins as well as potential proton pump inhibitors. However, most pH-detecting assays available are either not quantitative, require well-adapted reconstitution protocols or are not appropriate for single vesicle studies. Here, we describe the quantitative and time-resolved detection of F-type ATPase-induced pH changes across vesicular membranes doped with the commercially available pH sensitive fluorophore Oregon Green 488 DHPE. This dye is shown to be well suited to monitor acidification of lipid vesicles not only in bulk but also at the single vesicle level. The pKa value of Oregon Green 488 DHPE embedded in a lipid environment was determined to be 6.1 making the fluorophore well suited for a variety of physiologically relevant proton pumps. The TFOF1-ATPase from a thermophilic bacterium was reconstituted into large unilamellar vesicles and the bulk acidification assay clearly reveals the overall activity of the F-type ATPase in the vesicle ensemble with an average pH change of 0.45. However, monitoring the pH changes in individual vesicles attached to a substrate demonstrates that the fraction of vesicles with a significant observable pH change is only about 5%, a number that cannot be gathered from bulk experiments and which is considerably lower than expected.


 Please wait while we load your content...
Please wait while we load your content...