Ionic conductivity of acceptor doped sodium bismuth titanate: influence of dopants, phase transitions and defect associates†
Abstract
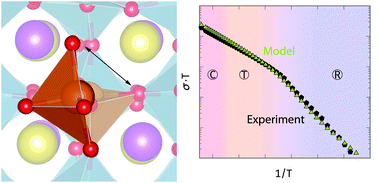
We investigate both, experimentally and theoretically, the electrical conductivity of Mg- and Fe-doped polycrystalline Na0.5Bi0.5TiO3. Samples with up to 4% of acceptor dopants are studied by means of impedance spectroscopy, scanning electron microscopy, and X-ray diffraction, while an analytical defect chemical model is developed for describing the measured conductivities. Within the framework of defect chemistry, we demonstrate that the experimentally measured conductivities can only be reproduced, if the formation of dopant–vacancy defect complexes is considered and the phase transition from a rhombohedral to a tetragonal symmetry is taken into account, affecting the dissociation of the dopant–vacancy complex. By using migration energies from density functional theory calculations, we obtain a good agreement between the data obtained from the analytical model and the experimental results, if we assume that the association energy is strongly affected by the dopant concentration.


 Please wait while we load your content...
Please wait while we load your content...