H2O-induced self-propagating synthesis of hierarchical porous carbon: a promising lithium storage material with superior rate capability and ultra-long cycling life†
Abstract
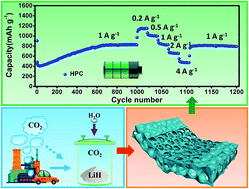
Hierarchical porous carbon (HPC) has attracted much attention in tackling global environmental and energy problems. For the state-of-the-art routes to synthesize HPC from organic compounds, the emission of carbon dioxide (CO2) and gaseous pollutants is inevitable during thermal carbonization. Herein, we report an environmentally benign and high-yield route to synthesize HPC from CO2via H2O-induced self-propagating reactions. By introducing an initiator of H2O, CO2 can react with lithium hydride (LiH) to produce HPC in 13 seconds at low temperatures. The as-synthesized HPC exhibits an interconnected micro–meso–macropore network structure with a high porosity of 83%. The formation mechanism of HPC is discussed on the basis of the conversion reactions from CO2 to C and the gas blowing effect in producing hierarchical porosity. The HPC evaluated as an anode material for lithium-ion batteries not only delivers a high reversible capacity of ∼1150 mA h g−1 at a current density of 0.2 A g−1, but also exhibits superior rate capability (∼825 mA h g−1 at 1.0 A g−1) and excellent cycling properties (up to 2000 cycles). This research opens a new avenue both to synthesize HPC from CO2 on a large scale and to mitigate greenhouse gas from the atmosphere.


 Please wait while we load your content...
Please wait while we load your content...