Unraveling factors leading to efficient norbornadiene–quadricyclane molecular solar-thermal energy storage systems†
Abstract
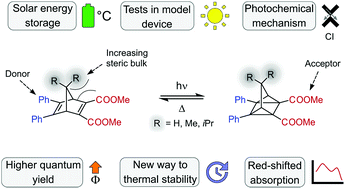
Developing norbornadiene–quadricyclane (NBD–QC) systems for molecular solar-thermal (MOST) energy storage is often a process of trial and error. By studying a series of norbornadienes (NBD-R2) doubly substituted at the C7-position with R = H, Me, and iPr, we untangle the interrelated factors affecting MOST performance through a combination of experiment and theory. Increasing the steric bulk along the NBD-R2 series gave higher quantum yields, slightly red-shifted absorptions, and longer thermal lifetimes of the energy-rich QC isomer. However, these advantages are counterbalanced by lower energy storage capacities, and overall R = Me appears most promising for short-term MOST applications. Computationally we find that it is the destabilization of the NBD isomer over the QC isomer with increasing steric bulk that is responsible for most of the observed trends and we can also predict the relative quantum yields by characterizing the S1/S0 conical intersections. The significantly increased thermal half-life of NBD-iPr2 is caused by a higher activation entropy, highlighting a novel strategy to improve thermal half-lives of MOST compounds and other photo-switchable molecules without affecting their electronic properties. The potential of the NBD-R2 compounds in devices is also explored, demonstrating a solar energy storage efficiency of up to 0.2%. Finally, we show how the insights gained in this study can be used to identify strategies to improve already existing NBD–QC systems.


 Please wait while we load your content...
Please wait while we load your content...