Side-chain engineering in a thermal precursor approach for efficient photocurrent generation†
Abstract
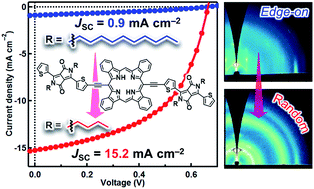
An ideal active-layer compound for bulk-heterojunction (BHJ) organic photovoltaic devices (OPVs) can assemble upon deposition to form the effective π–π stacking that facilitates exciton diffusion and charge-carrier transport. It is also expected to possess high-enough miscibility for forming sufficient heterojunctions to ensure efficient charge separation. However, these characteristics are often not compatible in organic small-molecule semiconductors: compounds endowed with rich self-π–π interaction capacity tend to be poor in miscibility, or maybe even insoluble in extreme cases. Herein, we postulate that a thermal precursor approach can serve as a way out of this dilemma, provided that molecules are properly engineered. This work evaluates a series of diketopyrrolopyrrole (DPP)–tetrabenzoporphyrin (BP) conjugates named Cn-DPP–BP (n = 4, 6, 8 or 10 depending on the length of alkyl groups on the DPP unit) as a p-type material in BHJ OPVs. These compounds are strongly aggregating and insoluble, thus processed via the thermal precursor approach in which the corresponding soluble derivatives (Cn-DPP–CP) are solution-processed into thin films and then converted to the target materials by in situ thermal reactions. The comparative study shows that the short-circuit current density largely depends on the length of alkyl substituents, ranging from 0.88 mA cm−2 with C10-DPP–BP to 15.2 mA cm−2 with C4-DPP–BP. Investigation into the structure of active layers through fluorescence-decay analysis, atomic-force microscopy, and two-dimensional grazing-incidence wide-angle X-ray diffractometry indicates that the introduction of shorter alkyl chains positively affects the miscibility and molecular orientation in BHJ layers. This trend is not fully parallel to those observed in the BHJ systems prepared through conventional solution techniques, and will provide a unique basis for devising a new class of high-performance OPV materials.


 Please wait while we load your content...
Please wait while we load your content...