Inverse relationship of dimensionality and catalytic activity in CO2 transformation: a systematic investigation by comparing multidimensional metal–organic frameworks†
Abstract
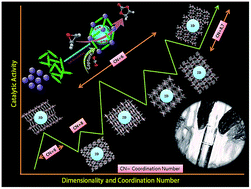
The correlation between dimensionality and active sites on deciding the catalytic performance of an MOF catalyst in CO2–epoxide cycloaddition reactions has been studied. Seven In(III) based MOFs built from carboxylic and N-donor ligands possessing different dimensionalities and distinct coordination environments were chosen as solid acid catalysts for this study. The origin of the catalytic activity of an In3+/TBAB bifunctional system in a CO2–PO reaction was studied in detail by performing density functional theory (DFT) calculations at the M06/LACVP**++ level. The energy barrier of the propylene oxide ring opening in the presence of In3+/Br− is 11.5 kcal mol−1, which is significantly lower than those of un-catalyzed (55–63 kcal mol−1) and Br−-catalyzed (19.5 kcal mol−1) reactions, which confirms the importance of the In3+/Br− binary catalytic system in the CO2–epoxide cycloaddition reactions. The one-dimensional (1D) MOF with unsaturated metal centers exhibited higher catalytic activity (PO conversion: 91%, temperature: 50 °C, and time: 12 h) than the two- and three-dimensional MOFs. The roles of dimensionality and unsaturated metal centers in cycloaddition reactions were explained on the basis of the results of activity testing and structural investigations. In addition, a plausible reaction mechanism for the catalytic activity of the 1D MOF was proposed with reference to our structure-density functional theory correlations.


 Please wait while we load your content...
Please wait while we load your content...