Dual-functional CoAl layered double hydroxide decorated α-Fe2O3 as an efficient and stable photoanode for photoelectrochemical water oxidation in neutral electrolyte†
Abstract
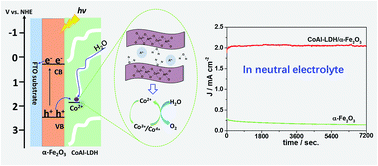
As a promising photoanode material, hematite (α-Fe2O3) can be used for photoelectrochemical (PEC) water oxidation but there are still great challenges, such as low efficiency and poor stability in neutral pH electrolyte, before it can be used in practice. Herein, a CoAl layered double hydroxide (CoAl-LDH) has been deposited in situ onto α-Fe2O3 nanoarrays. The PEC performances of the resulting composite photoanodes (CoAl-LDH/α-Fe2O3) towards water oxidation were investigated in neutral pH electrolyte. Contrastive experiments indicated that Co2+ in CoAl-LDH provided active catalytic sites for water oxidation, while Al3+ was inactive and offered support for the layered skeleton. CoAl-LDH could have a dual role as an efficient cocatalyst to lower the onset potential and enhance the photocurrent of α-Fe2O3, and as an effective protector of α-Fe2O3 from corrosion in neutral pH electrolyte. Relative to that of bare α-Fe2O3, a ∼250 mV negatively shifted onset potential and an almost 9-fold enhancement in photocurrent density at 1.23 V vs. RHE were observed with CoAl-LDH/α-Fe2O3. Moreover, CoAl-LDH/α-Fe2O3 showed excellent stability at 1.23 V vs. RHE with a steady photocurrent of ∼2 mA cm−2 even after 2 h of irradiation, which was very close to 100% of the initial value. Electrochemical impedance spectroscopy analysis revealed that the charge transfer resistance at the interface of α-Fe2O3/electrolyte was significantly decreased by CoAl-LDH decoration. Using H2O2 as a hole scavenger, the charge injection efficiency at the interface of CoAl-LDH/α-Fe2O3 and electrolyte was recorded as 84% at 1.23 V vs. RHE, which was 4-fold higher than that at the interface of α-Fe2O3 and electrolyte. This work demonstrates that CoAl-LDH decorated semiconductors have great potential as efficient and stable photoanodes towards high PEC performance in neutral pH electrolyte.


 Please wait while we load your content...
Please wait while we load your content...