Constructing magnetic Si–C–Fe hybrid microspheres for room temperature nitroarenes reduction†
Abstract
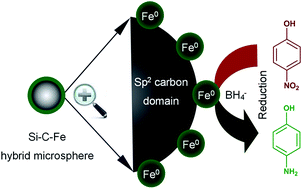
In this work, we present, for the first time, the synthesis and characterization of magnetic Si–C–Fe hybrid microspheres and their catalytic performance in room temperature reduction of 4-nitrophenol as a representative sustainable process for converting environmental pollutants to fine chemicals. The ferrocene-modified polydivinylbenzene (Fc-PDVB) precursor was synthesized by Pt-catalyzed hydrosilylation between the residual vinyl groups on the PDVB surface and 1,1′-bis (dimethylsilyl)ferrocene, where further pyrolysis led to the formation of Fe nanocrystal-containing Si–C–Fe hybrid microspheres. The precursor and hybrid microspheres were characterized by transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FT-IR), BET surface area/porosity, powder X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), magnetic properties and MAS solid-state NMR measurements. The ultimate microspherical catalyst exhibited nano- and meso-pores, a high specific surface area (i.e., 347.9 m2 g−1) and good ferromagnetic properties. Efficient catalytic activity (TOF: 0.163 s−1), 100% selectivity (to 4-aminophenol) and excellent reusability (with easy separation) have been delivered. The achieved microspheres outperform a number of nanomaterials such as supported noble metal particles, composites, monoliths and sheets. We have confirmed by DFT calculations that the activation of 4-nitrophenol via its weak non-covalent interaction with the sp2 carbon domain of Si–C–Fe hybrid microspheres contributed to the superior performance which can be extended to a range of nitrobenzenes.


 Please wait while we load your content...
Please wait while we load your content...