Three-dimensional π-conjugated compounds as non-fullerene acceptors in organic photovoltaics: the influence of acceptor unit orientation at phase interfaces on photocurrent generation efficiency†
Abstract
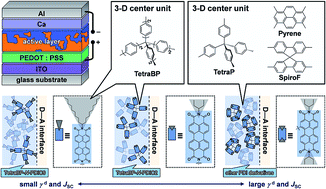
Recently, organic photovoltaics (OPVs) using electron-accepting π-conjugated systems as non-fullerene acceptors have been extensively studied. The fine-tuning of donor–acceptor (D–A) interfaces in bulk-heterojunction (BHJ) structures is crucial for accomplishing high power conversion efficiencies (PCEs); however, the rational design of non-fullerene acceptors for control over the film morphology is still unclear. To investigate the influence of structural modification on D–A interfaces, we synthesized a series of three-dimensional (3D) π-conjugated acceptors that contain perylene bis(dicarboximide) (PDI) units. These compounds showed little difference in the molecular properties. However, OPVs containing a blend of the poly[[4,8-bis[(2-ethylhexyl)oxy]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl][3-fluoro-2-[(2-ethylhexyl)carbonyl]thieno[3,4-b]thiophenediyl]] (PTB7) donor and an acceptor showed different PCEs, ranging from 0.02 to 2.02%, and originating from the differences in the short-circuit current densities (JSC). By investigating the blended film properties, we found that the degree of charge-separation mainly influences the photovoltaic characteristics of the OPVs. Furthermore, the JSC of OPVs and the London dispersion (γd) components of the surface free energy of the 3D acceptors are correlated. Consequently, increasing the interfacial exposure of the π-conjugated framework increases the value of γd orienting the PDI π-planes toward the D–A interfaces, which is desirable for the efficient charge separation into free carriers. This study highlights the importance of γd for the molecular design of non-fullerene acceptors for BHJ-type OPVs.


 Please wait while we load your content...
Please wait while we load your content...