Pt-like electrocatalytic behavior of Ru–MoO2 nanocomposites for the hydrogen evolution reaction†
Abstract
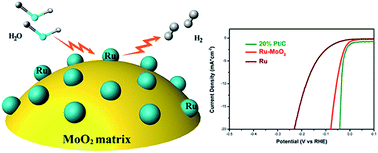
The design and development of inexpensive highly efficient electrocatalysts are critically important for their practical applications in the hydrogen evolution reaction (HER). Plain Ru and MoO2 are not very active for the HER under alkaline conditions. In this work, we report the preparation of Ru–MoO2 nanocomposites via a facile in situ carburization of a Ru modified Mo-based metal–organic framework. The nanocomposites exhibited very low overpotential and superior stability to achieve 10 mA cm−2 under both acidic and alkaline conditions (55 mV in 0.5 M H2SO4 and 29 mV in 1 M KOH). Their excellent performance under alkaline conditions was even better than that of 20% Pt/C. Our experimental and computational (DFT) results reveal that the remarkable activity stems from the synergistic interplay produced by strong electronic interactions between MoO2 and Ru nanoparticles. This modulated electronic structure accompanying enhanced electrical conductivity would significantly improve the catalytic activity. This strategy provides an insight into the design and synthesis of a low-cost and high performance alternative to Pt-based catalysts for the HER.

 Please wait while we load your content...
Please wait while we load your content...