Polymorphism in the family of Ln6−xMoO12−δ (Ln = La, Gd–Lu; x = 0, 0.5) oxygen ion- and proton-conducting materials
Abstract
The formation of Ln6−xMoO12−δ (Ln = La, Gd, Dy, Ho, Er, Tm, Yb, Lu; x = 0, 0.5) rare-earth molybdates from mechanically activated oxide mixtures has been studied in the range 900–1600 °C. The morphotropy and polymorphism (thermodynamic phase and kinetic (growth-related) transitions) of the Ln6−xMoO12−δ (Ln = La, Gd–Lu; x = 0, 0.5) molybdates have been analyzed in detail. As a result we have observed two new types of oxygen ion- and proton-conducting materials with bixbyite (Ia![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) , no. 206) and rhombohedral (R
, no. 206) and rhombohedral (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) , no. 148) structures in the family of Ln6−xMoO12−δ (Ln = La, Gd–Lu; x = 0, 0.5) molybdates. The heavy rare-earth molybdates Ln6−xMoO12−δ (Ln = Er, Tm, Yb; x = 0, 0.5) have been shown for the first time to undergo an order–disorder (rhombohedral–bixbyite) phase transition at 1500–1600 °C, and we have obtained compounds and solid solutions with the bixbyite structure (Ia
, no. 148) structures in the family of Ln6−xMoO12−δ (Ln = La, Gd–Lu; x = 0, 0.5) molybdates. The heavy rare-earth molybdates Ln6−xMoO12−δ (Ln = Er, Tm, Yb; x = 0, 0.5) have been shown for the first time to undergo an order–disorder (rhombohedral–bixbyite) phase transition at 1500–1600 °C, and we have obtained compounds and solid solutions with the bixbyite structure (Ia![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ). The stability range of the rhombohedral phase (R
). The stability range of the rhombohedral phase (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) increases with decreasing Ln ionic radius across the Ln6−xMoO12−δ (Ln = Er, Tm, Yb, Lu) series. We have detected a proton contribution to the conductivity of the rhombohedral La5.5MoO11.25 (2 × 10−4 S cm−1 at 600 °C in wet air) and high-temperature polymorph Yb6MoO12−δ bixbyite structure, (Ia
) increases with decreasing Ln ionic radius across the Ln6−xMoO12−δ (Ln = Er, Tm, Yb, Lu) series. We have detected a proton contribution to the conductivity of the rhombohedral La5.5MoO11.25 (2 × 10−4 S cm−1 at 600 °C in wet air) and high-temperature polymorph Yb6MoO12−δ bixbyite structure, (Ia![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) below 600 °C. At these temperatures, rhombohedral (R
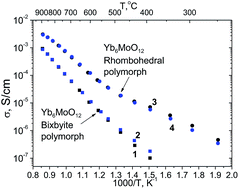
) below 600 °C. At these temperatures, rhombohedral (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) Yb6MoO12 seems to be an oxygen ion conductor (Ea = 0.53–0.58 eV). The total conductivity of rhombohedral (R
) Yb6MoO12 seems to be an oxygen ion conductor (Ea = 0.53–0.58 eV). The total conductivity of rhombohedral (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) Yb6MoO12 exceeds that of bixbyite Yb6MoO12−δ by more than one order of magnitude and is 3 × 10−5 S cm−1 at 500 °C. According to their high-temperature (T > 600 °C) activation energies, the lanthanum and ytterbium molybdates studied here are mixed electron–ion conductors.
) Yb6MoO12 exceeds that of bixbyite Yb6MoO12−δ by more than one order of magnitude and is 3 × 10−5 S cm−1 at 500 °C. According to their high-temperature (T > 600 °C) activation energies, the lanthanum and ytterbium molybdates studied here are mixed electron–ion conductors.


 Please wait while we load your content...
Please wait while we load your content...