Photodeposited ruthenium dioxide films for oxygen evolution reaction electrocatalysis†
Abstract
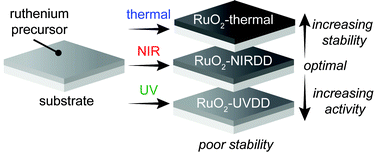
Ruthenium dioxide (RuO2) is an excellent catalyst for a wide range of electrosynthesis applications, including the oxygen evolution reaction (OER). Herein we describe a facile approach for accessing ruthenium dioxide films that produce high OER electrocatalytic activities in acidic media. Exposure of spin-cast ruthenium trichloride films on fluorine-doped SnO2-coated glass (FTO) substrates to near-infrared radiation, “near-infrared driven decomposition (NIRDD)”, induces a thermal decomposition process that affords nanocrystalline ruthenium dioxide. In contrast, ultraviolet-driven-decomposition (UVDD) of the ruthenium precursor Ru(tmhd)2(COD) [tmhd = bis(2,2,6,6-tetramethyl-3,5-heptanedionato); COD = 1,5-cyclooctadiene] affords an amorphous oxide film. The amorphous RuO2 films suffer from poor adhesion to FTO, whereas the nanocrystalline RuO2 films display electrochemical properties that match those exhibited by state-of-the-art RuO2-based OER electrocatalysts (η ∼ 360 mV at 10 mA cm−2 in 1 M H2SO4) and show stabilities that are consonant with crystalline films.


 Please wait while we load your content...
Please wait while we load your content...