Improved electro-grafting of nitropyrene onto onion-like carbon via in situ electrochemical reduction and polymerization: tailoring redox energy density of the supercapacitor positive electrode†
Abstract
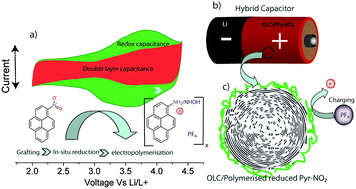
Herein, we report an improved method for the physical grafting of 1-nitropyrene (Pyr-NO2) onto highly graphitized carbon onion. This is achieved through a lowering of the onset potential of the pyrene polymerization via in situ reduction of the NO2 group. The additional redox activity pertaining to the reduced NO2 enables exceeding the faradaic capacity, which is associated with the p-doping of the grafted pyrene backbone, as observed for pyrene, 1-aminopyrene, and unreduced Pyr-NO2. Theoretical calculations demonstrate the charge transfer and binding enthalpy capabilities of Pyr-NO2, which are significantly higher than those of the other two species, and which allow for improved p-stacking on the carbon surface. Upon 20 wt% grafting of Pyr-NO2, the capacity of the electrode jumps from 20 mA h gelectrode−1 to 38 mA h gelectrode−1, which corresponds to 110 mA h g−1 per mass of Pyr-NO2 and the average potential is increased by 200 mV. Very interestingly, this high performance is also coupled with outstanding retention with respect to both the initial capacity for more than 4000 cycles, as well as the power characteristics, demonstrating the considerable advantages of employing the present in situ grafting technique.


 Please wait while we load your content...
Please wait while we load your content...