Two linkers are better than one: enhancing CO2 capture and separation with porous covalent triazine-based frameworks from mixed nitrile linkers†
Abstract
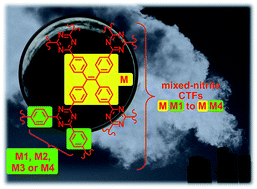
Covalent triazine-based framework (CTF) materials were synthesized by combining two different nitrile building blocks: the tetranitrile tetrakis(4-cyanophenyl)ethylene (M) was reacted with either terephthalonitrile (M1), tetrafluoroterephthalonitrile (M2), 4,4′-biphenyldicarbonitrile (M3) or 1,3,5-benzenetricarbonitrile (M4) under ionothermal conditions (ZnCl2, 400 °C) to yield mixed-nitrile MM′-CTFs MM1 to MM4. Comparative 1H/13C and 19F/13C CP MAS analyses of MM2(300) (synthesized at 300 °C) suggest that the hydrogenated and fluorinated carbon atoms are in close vicinity (<5 Å) to each other and support the formulation of the MM2(300) sample as a copolymeric CTF. Systematic N2, CO2 and CH4 gas sorption studies were performed up to 1 bar at 273 K and 293 K. The specific BET surface areas of MM1–MM4 were 1800, 1360, 1884 and 1407 m2 g−1, respectively. The CO2 uptake capacity of mixed-nitrile MM1, MM2 and MM4 was higher than the CO2 uptake of the respective individual single-nitrile M- or M′-CTF despite a higher surface area of the M-CTF PCTF-1 (2235 m2 g−1). The synergistic increase in the CO2 uptake of the mixed-nitrile MM′-CTFs is due to the higher CO2-accessible micropore volume Vmicro(CO2) and the higher micropore volume fraction V0.1/Vtot of the MM′-CTFs compared to the M- or M′-CTFs. The surface area of porous materials does not play the most important role in CO2 storage at low pressure but the CO2-accessible micropore volume is the more decisive factor. Further, MM2 shows the second highest (of known CTFs synthesized at 400 °C) CO2 uptake capacity of 4.70 mmol g−1 at 273 K and 1 bar because of its large micropore fraction (82%), which may be due to the release of fluorous decomposition products (‘defluorination carbonization’) during its synthesis. The CO2/N2 adsorption selectivities of mixed-nitrile MM1, MM2 and MM4 CTFs were also higher than those of the single-nitrile component M- or M′-CTFs.


 Please wait while we load your content...
Please wait while we load your content...