Phase transfer of TiO2 nanoparticles from water to ionic liquid triggered by phosphonic acid grafting†
Abstract
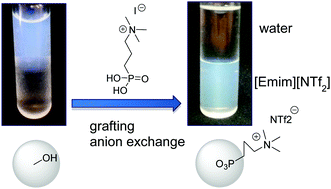
Controlling the interface between TiO2 nanocrystals and ionic liquids is of high fundamental and applied interest for energy storage and conversion devices. Phase transfer of nanoparticles from a synthesis medium to a processing or an application medium plays a significant role in nanotechnology. Here we demonstrate that surface modification with phosphonic acids bearing cationic end-groups can trigger the phase transfer of TiO2 nanoparticles from an aqueous sol to a typical water-immiscible ionic liquid, [Emim][NTf2]. The transfer involves both the grafting of the phosphonic acid moiety and the exchange of the counter ion of the cationic end-group by NTf2 anions, as demonstrated by solid-state NMR, elemental analysis and independent grafting and ion exchange experiments. Furthermore, the colloidal stability of the TiO2 sols in [Emim][NTf2] strongly depends on the hydrophobic character of the cationic end-groups.


 Please wait while we load your content...
Please wait while we load your content...