Evaporated manganese films as a starting point for the preparation of thin-layer MnOx water-oxidation anodes†
Abstract
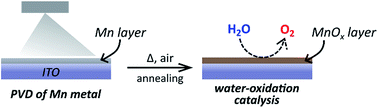
A novel method to prepare anodes for water electrolysis cells has been developed, which starts from layers of elemental manganese deposited by physical vapour deposition (PVD) on indium-doped tin oxide (ITO). Oxidation in dry air at 300 °C transforms this metallic Mn layer into a manganese(II)-rich MnOx coating (x = 1–1.3), which also contains a buried layer of an In–Sn alloy originating from reactions with the ITO support. The MnOx films are well connected to the underlying substrate and act as efficient catalysts for water-oxidation catalysis (WOC) at neutral pH. Detailed post-operando analyses using XRD, SEM, TEM and XAS revealed that the dense MnO/Mn3O4 film is virtually not affected by 2 h of electrochemical WOC at E ≈ +1.8 V vs. RHE, corresponding well to the observed good stability of catalytic currents, which is unusual for such thin layers of a MnOx catalyst. The current densities during electrolyses are so far low (i ≈ 50–100 μA cm−2 at pH 7), but optimization of the preparation process may allow for significant improvements. This new, rather easy, and adaptable preparation method for stable, thin-layer MnOx water-oxidation anodes could thus prove to be very useful for a variety of applications.
- This article is part of the themed collection: 3rd International Solar Fuels Conference and International Conference on Artificial Photosynthesis


 Please wait while we load your content...
Please wait while we load your content...