In situ observation of reactive oxygen species forming on oxygen-evolving iridium surfaces†
Abstract
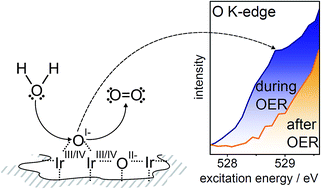
Water splitting performed in acidic media relies on the exceptional performance of iridium-based materials to catalyze the oxygen evolution reaction (OER). In the present work, we use in situ X-ray photoemission and absorption spectroscopy to resolve the long-standing debate about surface species present in iridium-based catalysts during the OER. We find that the surface of an initially metallic iridium model electrode converts into a mixed-valent, conductive iridium oxide matrix during the OER, which contains OII− and electrophilic OI− species. We observe a positive correlation between the OI− concentration and the evolved oxygen, suggesting that these electrophilic oxygen sites may be involved in catalyzing the OER. We can understand this observation by analogy with photosystem II; their electrophilicity renders the OI− species active in O–O bond formation, i.e. the likely potential- and rate-determining step of the OER. The ability of amorphous iridium oxyhydroxides to easily host such reactive, electrophilic species can explain their superior performance when compared to plain iridium metal or crystalline rutile-type IrO2.
- This article is part of the themed collection: Global Energy Challenges: Electrochemical Energy

 Please wait while we load your content...
Please wait while we load your content...