Enzyme-triggered compound release using functionalized antimicrobial peptide derivatives†
Abstract
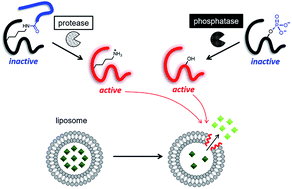
Controlled release is one of the key technologies for medical innovation, and many stimulus-responsive nanocarriers have been developed to utilize this technology. Enzyme activity is one of the most useful stimuli, because many enzymes are specifically activated in diseased tissues. However, controlled release stimulated by enzyme activity has not been frequently reported. One of the reasons for this is the lack of versatility of carriers. Most of the reported stimulus-responsive systems involve a sophisticated design and a complicated process for the synthesis of stimulus-responsive nanocarrier components. The purpose of this study was to develop versatile controlled release systems triggered by various stimuli, including enzyme activity, without modifying the nanocarrier components. We developed two controlled release systems, both of which comprised a liposome as the nanocarrier and a membrane-damaging peptide, temporin L (TL), and its derivatives as the release-controllers. One system utilized branched peptides for proteases, and the other utilized phosphopeptides for phosphatases. In our systems, the target enzymes converted the non-membrane-damaging TL derivatives into membrane-damaging peptides and released the liposome inclusion. We demonstrated the use of our antimicrobial peptide-based controlled release systems for different enzymes and showed the promise of this technology as a novel theranostic tool.


 Please wait while we load your content...
Please wait while we load your content...