Recent developments in and perspectives on three-coordinate boron materials: a bright future
Abstract
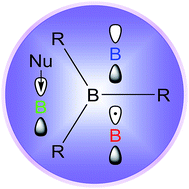
The empty pz-orbital of a three-coordinate organoboron compound leads to its electron-deficient properties, which make it an excellent π-acceptor in conjugated organic chromophores. The empty p-orbital in such Lewis acids can be attacked by nucleophiles, so bulky groups are often employed to provide air-stable materials. However, many of these can still bind fluoride and cyanide anions leading to applications as anion-selective sensors. One electron reduction generates radical anions. The π-acceptor strength can be easily tuned by varying the organic substituents. Many of these compounds show strong two-photon absorption (TPA) and two-photon excited fluorescence (TPEF) behaviour, which can be applied for e.g. biological imaging. Furthermore, these chromophores can be used as emitters and electron transporters in OLEDs, and examples have recently been found to exhibit efficient thermally activated delayed fluorescence (TADF). The three-coordinate organoboron unit can also be incorporated into polycyclic aromatic hydrocarbons. Such boron-doped compounds exhibit very interesting properties, distinct from their all-carbon analogues. Significant developments have been made in all of these areas in recent years and new applications are rapidly emerging for this class of boron compounds.
- This article is part of the themed collections: Celebrating 15 years of exceptional research in Chemical Science and Most downloaded articles of 2017: Inorganic and Physical Chemistry


 Please wait while we load your content...
Please wait while we load your content...