Targeted synthesis of cadmium(ii) Schiff base complexes towards corrosion inhibition on mild steel†
Abstract
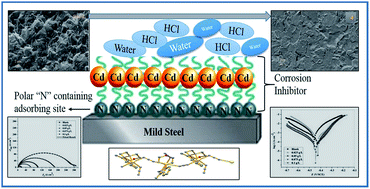
Three ligands L1 [N,N-dimethyl-N′-(1-pyridin-2-yl-ethylidene)-ethane-1,2-diamine], L2 [2-morpholino-N-(1-(pyridin-2-yl)ethylidene)ethanamine] and L3 [(2-(piperidin-1-yl)-N-(1-(pyridin-2-yl)ethylidene)ethanamine)] were introduced to prepare five Cd(II) Schiff base complexes [Cd(L1)2](ClO4)2 (1), [Cd(L1)(Cyanoacetate)(OAc)] (2), [Cd2(L1)2(N3)4] (3), [Cd(L2)(N3)2]n (4), [Cd2 (L3)2(N3)4]n (5) and the corrosion inhibition properties of these complexes on mild steel upon treatment with 15% HCl have been examined where azide complexes have shown corrosion inhibition properties as revealed by electrochemical impedance spectroscopy and potentiodynamic polarization. Field emission scanning electron microscopy (FE-SEM) images show that the mild steel surface was protected by cadmium complexes. Among azido complexes polymeric complexes have higher inhibition activity which was further explained using density functional theory. So finally a new bridge between nuclearity driven coordination inorganic chemistry and materials as well as corrosion engineering was established by this observation.


 Please wait while we load your content...
Please wait while we load your content...