Rational fabrication of a graphitic-C3N4/Sr2KNb5O15 nanorod composite with enhanced visible-light photoactivity for degradation of methylene blue and hydrogen production†
Abstract
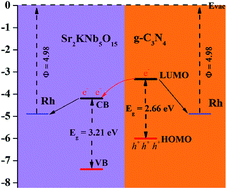
A g-C3N4/Sr2KNb5O15 nanorod composite photocatalyst was simply prepared by direct growth of graphitic C3N4 on one-dimensional Sr2KNb5O15 nanorods and evaluated by degradation of methylene blue (MB) and water splitting for H2 production under visible light irradiation. By coupling g-C3N4 and Sr2KNb5O15 nanorods, the nanocomposite with an optimal 77 wt% g-C3N4 exhibited 6.2 times higher activity for photodegradation than the bare g-C3N4 and the increase in H2 production rate from an aqueous methanolic solution reached up to 12.1 fold after appropriate photodeposition of 0.03 wt% Rh cocatalysts. Furthermore, a comparative study on the preparation methods shows the photodegradation rate of the nanocomposite prepared by the direct growth method was found to be 3.4 times higher than those formed just by physical mixing. The results demonstrate the importance of the formation of proper nano-interfaces in the nanocomposite. Combined with theoretical prediction of the band structures, a possible mechanism is thus proposed including the formation of proper interfaces between g-C3N4 and Sr2KNb5O15 by direct growth approachs promoting spatial separation of photoinduced electron–hole pairs.


 Please wait while we load your content...
Please wait while we load your content...