γ-Al2O3 nanorods with tuneable dimensions – a mechanistic understanding of their hydrothermal synthesis†
Abstract
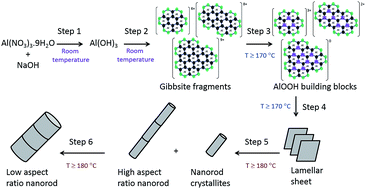
This paper reports for the first time the size control of well-defined and morphologically pure alumina (γ-Al2O3) nanorods, presenting an economic and reproducible route for the manufacture of these materials with tuneable sizes for useful applications, for example serving as adsorbents, catalysts and catalyst supports. A detailed understanding of the different steps taking place during the hydrothermal synthesis has been deduced herein. Understanding the effect of temperature on the relative rates of these steps is essential for achieving size and morphology selectivity, but has often been overlooked in the literature. This systematic study identifies six distinct steps taking place during the synthesis: (1) formation of Al(OH)3, (2) dissolution of Al(OH)3 into hexameric based fragments (3) thermolysis at temperatures ≥ 170 °C into soluble AlOOH (boehmite) building blocks (4) formation of lamellar AlOOH sheets (5) scrolling into nanorod crystallites and subsequent oriented attachment into high aspect nanorods and (6) growth by Ostwald ripening to low aspect nanorods. The obtained AlOOH nanorods are converted into γ-Al2O3 with conservation of morphology by calcination at 500 °C. Nanorod formation (step 5) can only be achieved at temperatures ≥ 180 °C (after 20 hours). At 180 °C, growth of the rods (step 6) takes place simultaneously with their slow formation (step 5) leading to two distinct nanorod products with different aspect ratios. At higher temperatures (200 °C), the rate of formation (step 5) is fast, quickly reaching completion, allowing for substantial growth of the nanorods and resulting in a monomodal size distribution. Thus, we have identified that γ-Al2O3 rods with high aspect ratio can be selectively synthesised at 180 °C for ≥20 hours, while low aspect ratios are produced at 200 °C for ≥10 hours. In all cases, the average size of the nanorods increases linearly with prolonged reaction time due to their continuous growth.
- This article is part of the themed collection: Introducing the Reaction Chemistry & Engineering Associate Editors


 Please wait while we load your content...
Please wait while we load your content...