Synthesis and electrochemical performance of a polymer-derived silicon oxycarbide/boron nitride nanotube composite
Abstract
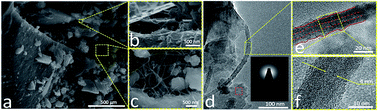
Polymer-derived ceramics (PDCs) such as silicon oxycarbide (SiOC) have shown promise as electrode materials for rechargeable Li-ion batteries (LIBs) owing to the synergy between their disordered carbon phase and hybrid bonds of silicon with oxygen and carbon. This study describes the synthesis and characterization of a new type of PDC composite comprising SiOC embedded with varying wt% of boron nitride nanotubes (BNNTs) and supported on reduced graphene oxide. Electron microscopy and a range of spectroscopic characterization techniques were employed to confirm the polymer-to-ceramic transformation and presence of a disordered carbon phase. The composites were tested as electrode materials for LIB and symmetric supercapacitor applications. As an electrode material for LIB, SiOC–BNNT paper composite containing 0.25 wt% BNNT demonstrated the highest charge capacity of 410 mA h g−1 (at 100 mA g−1) which stabilized at 238 mA h g−1. On the contrary, composite electrode with 0.5 wt% BNNT demonstrated the highest specific capacitance corresponding to 78.93 F g−1 at 1 A g−1 with a cyclic retention of 86% after 185 cycles. This study shows that presence of BNNTs in the composite influenced the free carbon phase in SiOC and its electrochemical performance. As such, the composite may be used as a flexible electrode material for electrochemical energy storage devices.


 Please wait while we load your content...
Please wait while we load your content...