Development of arginine based nanocarriers for targeting and treatment of intracellular Salmonella†
Abstract
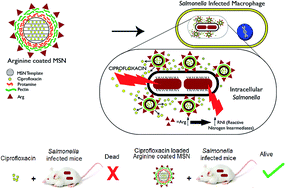
Arginine decorated nanocarriers exhibited intravacuolar targeting capability which was utilized to deliver antibiotics into the intracellular niche of pathogens like Salmonella and Mycobacterium. The arginine based nanocarrier system (Arg-MSN) was developed on a mesoporous silica nanoparticle (MSN) template by conjugating L-arginine to protamine and pectin coated MSN by using a layer-by-layer coating approach. The synthesized nanocarriers were characterized using microscopy, FTIR spectroscopy, and zeta potential analyses. Lower cytotoxicity and hemolysis was observed for Arg-MSN nanocarrier compared to bare MSN template. Ciprofloxacin, a fluoroquinolone antibiotic was entrapped in Arg-MSN which showed gradual release of ciprofloxacin over a period of 24 h. In vitro experiments in Salmonella infected macrophages and epithelial cells exhibited two-fold higher antibacterial activity with ciprofloxacin-loaded Arg-MSN (Cip Arg-MSN) compared to free ciprofloxacin. The increased antibacterial activity of Cip Arg-MSN is believed to result from co-localization of Arg-MSN with the intravacuolar Salmonella and localized delivery of the antibiotic. We also observe an increase in reactive nitrogen species upon Arg-MSN treatment in the infected cells. In vivo bacterial burden and morbidity studies exhibited nearly ten-fold lower Salmonella burden in the infected organs such as spleen, liver and MLN (mesenteric lymph nodes). Similar survival rates were observed at a lower dosage of Cip Arg-MSN over free ciprofloxacin. The coordinated effect of improved antibiotic delivery, intracellular targeting and production of reactive nitrogen species was found to result in enhanced antibacterial activity. The developed Arg-MSN system is expected to be an attractive carrier system for delivery of antibiotics for clearing intravacuolar infections.


 Please wait while we load your content...
Please wait while we load your content...