MOFs-Derived Co@CN bi-functional catalysts for selective transfer hydrogenation of α,β-unsaturated aldehydes without use of base additives†
Abstract
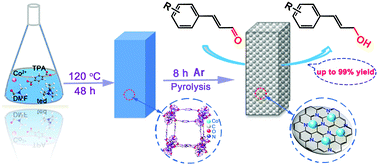
Transfer hydrogenation protocols involve versatile and environmentally benign methods capable of providing alternatives to traditional hydrogenation processes. However, most of the non-noble metal catalysts used for transfer hydrogenation processes proceed with the aid of base additives or phosphorus-containing ligands to obtain good yields. We report a green and sustainable catalytic system by fabrication of a Co@CN bi-functional nanocatalyst with a new N-containing [Co(TPA)(ted)0.5] as a sacrificial template. This catalytic system was employed effectively for selective transfer hydrogenation of an α,β-unsaturated aldehyde (cinnamaldehyde, CAL) to obtain a high yield of an α,β-unsaturated alcohol (cinnamyl alcohol, COL) under a mild reaction condition without use of additives. During thermolysis, the retained N atoms were distributed uniformly in catalysts and endowed the catalysts with abundant basic sites, and Co2+ in [Co(TPA)(ted)0.5] were converted in situ into Co nanoparticles. Experimental results indicated obvious coordination interactions between Co nanoparticles and N atoms in these bi-functional catalysts. By optimizing the reaction conditions, a catalytic system with Co@CN-900 as a catalyst and n-hexanol as the proton donor exhibited the optimal activity for this transformation, achieving 99% selectivity for COL with full conversion. In addition, a reasonable reaction mechanism for the transfer hydrogenation of CAL to COL was proposed. Furthermore, this catalytic system also demonstrated good recyclability as well as wide substrates compatibility.
- This article is part of the themed collection: Materials Chemistry Frontiers HOT articles for 2017

 Please wait while we load your content...
Please wait while we load your content...