Ring opening polymerization of macrolactones: high conversions and activities using an yttrium catalyst†
Abstract
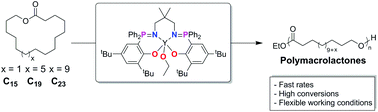
The ring-opening polymerization of macrolactones (C15–C23) enables the production of long-chain aliphatic polyesters which are crystalline polymers with melting temperatures ranging from 98–106 °C. Here, the polymerization of ω-pentadecalactone (C15), nonadecalactone (C19) and tricosalactone (C23) are investigated using an yttrium phosphasalen catalyst. The catalyst enables typical conversions to exceed >80% and the reactions occur either in neat monomer or in solution in toluene, over the temperature range 25–100 °C. The yttrium catalyst shows higher activities than previously reported aluminium–salen complexes, with TOF values in the range 200–400 h−1 in the best cases. The polymerizations occur with linear increase in molecular weight vs. conversion and enable the production of polyester with 10 < Mn< 60 kg mol−1. Using tricosalactone the polymerization thermodynamic parameters are determined and confirm the polymerization is entropically driven. The findings underscore the importance of continued catalyst development to allow higher rates of reaction which has the added benefit of accessing the highest conversions to polymer.


 Please wait while we load your content...
Please wait while we load your content...