Cationic peptidopolysaccharides synthesized by ‘click’ chemistry with enhanced broad-spectrum antimicrobial activities†
Abstract
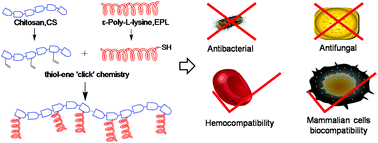
The emergence and spread of multidrug-resistant (MDR) bacteria is a serious clinical problem, and a challenge for the medical research community. There is an urgent need to explore novel effective antimicrobial molecules. Here, we report on a series of cationic peptidopolysaccharides that have a bacterial peptidoglycan-mimetic structure and have been synthesized using a facile thiol–ene ‘click’ chemistry to graft antimicrobial peptides, ε-poly-L-lysine (EPL), onto a polysaccharide (chitosan, CS) backbone. These CS-g-EPL copolymers demonstrated enhanced, selective, broad-spectrum antibacterial and antifungal activities towards Gram-negative bacteria (Escherichia coli and Pseudomonas aeruginosa), Gram-positive bacteria (Enterococcus faecalis and methicillin-resistant Staphylococcus aureus (MRSA)), and fungi (Candida albicans and Fusarium solani). Mechanistic studies proved that the CS-g-EPL likely disrupts the anionic microbial membrane leading to cell death, and is less likely to disrupt the mammalian cell membrane. In addition, CSL-g-EPL50% exhibits good in vitro biocompatibility with mammalian cells, and very little evidence of in vivo toxicity. Most importantly, CS-g-EPL demonstrated a strong antimicrobial efficacy with a log reduction of 4.66 in a rat MRSA infection model. These excellent biological properties present a promising prospect for CS-g-EPL in biomedical applications.


 Please wait while we load your content...
Please wait while we load your content...