Well-defined hyperstar copolymers based on a thiol–yne hyperbranched core and a poly(2-oxazoline) shell for biomedical applications†
Abstract
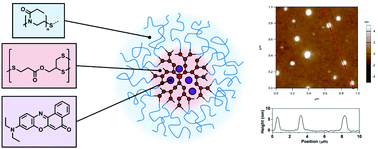
Well defined ‘hyperstar’ copolymers were synthesized by combining hyperbranched polymers produced by thiol–yne chemistry with poly(oxazoline)s. The hyperbranched core was prepared using an AB2 monomer and a trifunctional alkene, applying a monomer feeding approach. The degree of branching was high (0.9) while maintaining low dispersities (1.3). Poly(2-ethyl-2-oxazoline) (PEtOx) functionalized with a thiol end group was coupled to the surface of the hyperbranched structure accessing terminal alkyne units. PEtOx-SH was produced by the termination of the living polymerization with ethyl xanthate and subsequent conversion to thiol under alkaline conditions. The degree of polymerization was varied producing PEtOx with 23 or 42 repeating units, respectively with a dispersity of around 1.1. After conjugation of the polymer arms, hyperstar copolymers were characterized by SEC, NMR spectroscopy, light scattering, and AFM. The polymers were able to encapsulate the hydrophobic dye Nile red within the core of the structure with loading efficiencies between 0.3 and 0.9 wt%. Cytotoxicity of the hyperstars was assessed using A2780 human ovarian carcinoma cells resulting in IC50 values of around 0.7 mg ml−1. Successful internalization and colocalization with lysosomal compartments was observed by confocal microscopy studies.


 Please wait while we load your content...
Please wait while we load your content...