Multi-stimuli responsive block copolymers as a smart release platform for a polypyridyl ruthenium complex†
Abstract
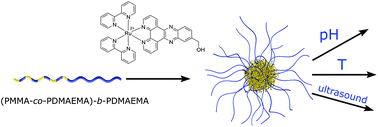
A variety of applications of amphiphilic block copolymers result from the control of their self-assembled structures. Herein, the synthesis and structure formation of block copolymers (BCPs) consisting of poly(N,N-dimethylaminoethyl methacrylate) (PDMAEMA) as one segment and poly(methyl methacrylate) (PMMA) or a statistical copolymer (PDMAEMA-co-PMMA) as a second segment, is described. The BCPs provide molar masses between 8.9 kg mol−1 and 35.6 kg mol−1 with low polydispersity index values, Đ = 1.05–1.13. BCPs are synthesized via sequential anionic polymerization strategies while structure formation in water is investigated by dynamic light scattering (DLS) and transmission electron microscopy (TEM). The PDMAEMA-containing micelles in water are loaded with a Ru(II) polypyridyl complex, i.e. [Ru(bipy)2-dppz-7-hydroxymethyl][PF6]2 (bipy = 2,2′-bipyridine; dppz = dipyridophenazine), which was previously shown to act as a potential photosensitizer in photodynamic therapy (PDT). Successful loading of the BCP micelles is evidenced by TEM measurements after dialysis in water. Stimulus-responsive release of the Ru(II) complex from the BCP micelles is shown using ultrasound, change of pH or temperature as external triggers. The quantification and release profiles for the Ru(II) complex are obtained by atomic absorption spectrometry (AAS). As a result, PDMAEMA-b-PMMA is not capable of releasing the Ru(II) complex in a controlled manner after application of, for instance, ultrasound or temperature change as external triggers due to the shielding (stealth effect) of the BCP. On the contrary, micelles made of BCPs featuring PDMAEMA and PDMAEMA-co-PMMA segments reveal excellent Ru(II) complex release profiles due to the tailored molecular composition of the underlying block segments as evidenced by temperature-dependent DLS and AAS measurements. Thus, these smart PDMAEMA-containing BCPs pave the way to a variety of applications for selective triggered release of small molecules.


 Please wait while we load your content...
Please wait while we load your content...