A study of cyclic carbonate aminolysis at room temperature: effect of cyclic carbonate structures and solvents on polyhydroxyurethane synthesis†
Abstract
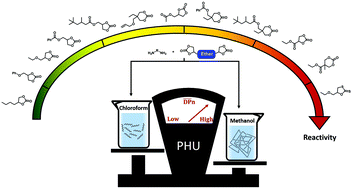
In recent years, intensive research and development have been carried out for the synthesis of isocyanate free polyurethanes from the reaction between five-membered cyclic carbonates and amines to yield polyhydroxyurethanes (PHU). Nevertheless, this reaction presents two main drawbacks: the low reactivity of cyclic carbonate aminolysis and the low molar masses of synthesized PHUs. Firstly, our work intends to propose a wider reactivity study on various C5 and C6 in order to determine the influence of substituents attached to the cyclic carbonate and to compare the reactivity of C5 and C6 with exactly the same substituents. Moreover, we completed this study with new structures with promising reactivity such as C5 and C6 with ethyl-ester or trimethylhexanoate substituents that have never been studied in the literature. Hence, a range of cyclic carbonate models with different structures, 5-, 6-membered cyclic carbonate (C5, C6) or 5-membered-dithio-cyclic carbonate (Cs), were synthesized. Secondly, this kinetic study has brought to light a partial conversion of precursors during the aminolysis reaction that can be responsible for the low molar masses of PHUs. This partial conversion is caused mainly by the formation of hydrogen bonding inter- and intra-chains limiting the diffusivity of species during the polymerization. A model study showed that the use of protic solvents improves the reactivity of cyclic carbonates and increases the conversion during their aminolysis reaction. Therefore, this protic solvent gives its mobility back to the precursors by limiting the creation of hydrogen bonding inter- and intra-chains. Finally, the efficiency of protic solvents on the synthesis of PHU polymers was evaluated.

 Please wait while we load your content...
Please wait while we load your content...